Dr. Webb has retired from the University of Rhode Island (July 2024) and will no longer be accepting graduate students or post-docs. She is continuing to publish and do research as Associate in Ichthyology at the Museum of Comparative Zoology at Harvard University.
This website will be updated going forward.
“What underlies great science is what underlies great art, whether it is visual or written, and that is the ability to distinguish patterns out of chaos” — Diana Gabaldon, author of Outlander
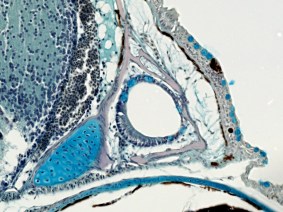
For several decades we have studied the structural and functional development and evolution of fish sensory systems, especially the mechanosensory lateral line system, a primitive vertebrate sensory system found in all 34,000+ fishes (and in larval and aquatic adult amphibians). The lateral line system detects water flows, which facilitates critical prey detection, predator avoidance, communication, rheotaxis, and navigation. Furthermore, unlike the nose, eyes and ears, which are bilateral sense organs found on the head, the lateral line system is composed of many small sense organs (neuromasts) located in arrays on the skin and in tubular canals on the head, trunk and tail. In bony fishes, canal neuromasts are found within a conserved subset of skull bones on the head and in the trunk canal in the lateral line scales on the body. Thus, the lateral line system has a dual identity – as a major component of the skull of bony fishes and as an essential sensory system that mediates critical behaviors. An understanding of the role of the lateral line system in behavior will also shed light on how fishes may overcome challenges in aquatic environments presented by global change.
Our recent work has been on the developmental and evolutionary sensory biology and ecology of freshwater tropical and temperate fishes, coral reef fishes, cichlid fishes, and deep sea fishes (see Research Page). Each taxon has interesting or unique attributes that have allowed us to ask fundamental questions about sensory system evolution, development, functional morphology, and the sensory basis for behavior. We use a suite of classic and cutting edge methods for the analysis of comparative and developmental morphology and behavior.
Recent Webb Lab News

- Dr. Webb was awarded the 2025 Joseph Nelson Award for Lifetime Achievement in Ichthyology by the American Society of Ichthyologists and Herpetologists
- Gus Bienenfeld (undergraduate in the Webb Lab) won Honorable Mention in the 2025 URI Research Photography competition for his image of a barb (a minnow) vitally stained to reveal the distribution of superficial neuromast organs on the head, trunk and tail.
- Dr. Webb was awarded the 2024 Robert K. Johnson Award for Excellence in Service by the American Society of Ichthyologists, of which she has been a member for 42 years (photo).
- Dr. Webb was awarded the 2024 URI Foundation Award for Teaching Excellence, the highest teaching honor at URI.
- Dr. Webb retired on June 30, 2024 and is now Professor Emerita at University of Rhode Island. She is continuing her research as an Associate in Ichthyology at the Museum of Comparative Zoology at Harvard University and through several collaborations.
Recent Research Highlights
-
- Marranzino, AN and Webb JF. (2025) “Photophores in stomiiform fishes: morphology, distribution and putative behavioral roles”. published in Biological Bulletin – describes the diversity of photophore organs among fishes in the largest order of exclusively deep sea fishes.
- Jones et al. (2025) “Salmonid sensory system development is affected by climate change driven temperature increases” (Jones, O’Donnell, Regish, and Webb) published in Scientific Reports – demonstrates the effect of increased temperature on the development of the LL system on the head in brook trout with implications for their ecology. Open Access.
- Chaumel et al., (2025) ” The tuna keel is a mechanosensory structure” (Chaumel, Wainwright, Webb, White, Lauder) published in iScience – describes the LL canal contained within the lateral keels of tunas, suggesting that sensory input to the neuromasts located in these canals may play a role in their locomotory behavior. Open Access.
- Jones et al., (2024b) “The Silverjaw Minnow, Ericymba buccata: An extraordinary lateral line system and its contribution to prey detection” (Jones, Maia, Conway, and Webb) – published in Integrative and Comparative Biology and based on an invited symposium talk at SICB – examines the anatomy of the unique dimorphic lateral line canal system of this species, describes the distribution of superficial neuromasts for the first time, and explores the sensory basis for prey detection.
- Jones et al. (2024a) “Development of the cranial lateral line system of Brook Trout, Salvelinus fontinalis (Teleostei: Salmonidae): Evolutionary and ecological implications”. (Jones, Rizzato, and Webb) published in Journal of Morphology – describes the development of the cranial LL canals and superficial neuromasts in brook trout and puts it into the context of the evolution and development of the LL system in basal teleost fishes.
- Webb (2023) – “Structural and functional evolution of the mechanosensory lateral line system of fishes” – Journal of the Acoustical Society of America (in special issue on Fish Bioacoustics) – invited research career retrospective that talks about the genesis of research in the Webb lab and the important contribution of serendipity in research.
- Hu et al. (2022) “Ear development in select coral reef fishes: Clues for the role of hearing in larval orientation behavior?” Ichthyology and Herpetology. 110: 759-775. Open Access. Awarded 2022 Best Paper in Ichthyology in Ichthyology and Herpetology (Young Investigator)
- Webb et al (2021) – members of the Webb and Maruska [LSU] labs published the first synthetic treatment of the anatomy, physiology and behavioral roles of the mechanosensory lateral line system of cichlid fishes. In: Abate ME & Noakes DLG (eds.). The Behavior, Ecology and Evolution of Cichlid Fishes: A Contemporary Modern Synthesis. Springer Academic, pp. 401-442.
- Majoris et al. (2021) – members of the Buston Lab (Boston U.), Webb Lab at URI and Paris Lab (U. Miami) published the first integrated study of ontogeny of multiple sensory systems and orientation behavior throughout the larval phase of any coral reef fish (a goby; Elacatinus lori). Open Access, in Scientific Reports.
- Nickles et al. (2020) “Organization and Ontogeny of a Complex Lateral Line System in a Goby (Elacatinus lori), with a Consideration of Function and Ecology” – in Copeia – provided the first detailed ontogeny of the lateral line system in any goby (the most speciose family of marine fishes and of coral reef fishes) revealing some of the “rules” underlying the organization of superficial neuromast proliferations in gobies and perhaps in other teleosts. Open Access. Awarded Best Paper in Ichthyology for 2020 in Copeia.
- Hu et al. (2018) “Potential roles of olfaction and taste in the orientation behavior of coral reef fish larvae: Insights from morphology”, published in Journal of Fish Biology. – Special Issue on Sensory Ecology of Fishes – analyzed the ontogeny of the nose and taste buds in pelagic larvae of several coral reef fishes (damselfishes, goby, cardinal fish) and used these data to suggest that taste, not (just?) olfaction, mediates the chemosensory cues that larvae may use in orienting towards coral reef settlement sites.
- Marranzino and Webb (2018) “Flow sensing in the deep sea: the lateral line system of stomiiform fishes“, published in Zoological Journal of the Linnean Society, revealed the presence of superficial neuromast proliferations in deep-sea stomiiform fishes for the first time, thus suggesting the importance of flow sensing in these ubiquitous and ecologically critical deep-sea fishes.
 Webb and Ramsay (2017) “A new interpretation of the 3-D configuration of lateral line scales and the lateral line canal contained within them“, published in Copeia, clarified incorrect figures published in textbooks and established a standard for understanding this fundamental feature of teleost (and other bony) fishes. Awarded Best Paper in Ichthyology for 2017 in Copeia.
Webb and Ramsay (2017) “A new interpretation of the 3-D configuration of lateral line scales and the lateral line canal contained within them“, published in Copeia, clarified incorrect figures published in textbooks and established a standard for understanding this fundamental feature of teleost (and other bony) fishes. Awarded Best Paper in Ichthyology for 2017 in Copeia.

